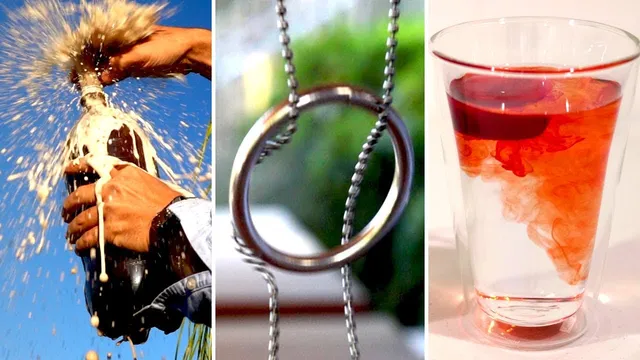
3 Perplexing Physics Problems
Based on Veritasium's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Shaking a carbonated drink doesn’t meaningfully raise headspace pressure; it mainly adds nucleation sites (tiny bubbles) in the liquid.
Briefing
A shaken carbonated drink doesn’t need a pressure boost to “explode”—it’s the sudden availability of tiny gas bubbles that turns a slow, equilibrium process into a rapid one. After a soda bottle sits for days at room temperature, the dissolved CO2 in the liquid is in equilibrium with the CO2 gas in the headspace, around 3 atmospheres (about 330 kPa). Shaking doesn’t meaningfully change that headspace pressure for typical bottles; instead, it introduces nucleation sites—microscopic air bubbles—into the liquid. When the bottle is opened and the external pressure drops to ~1 atmosphere, those bubbles expand and also provide surfaces where dissolved CO2 can come out of solution quickly. The result is a fast surge of fizz that can overflow the container.
That same theme—how hidden physical conditions control outcomes—runs through two other “perplexing” demonstrations. In the ice-melting challenge, identical ice cubes behave differently in fresh water versus saltwater: the cube in fresh water melts faster. The explanation hinges on density-driven convection. As the ice melts, the cold meltwater is denser than the surrounding fresh water, so it sinks and pulls warmer water down to the ice, accelerating melting. In saltwater, the cold meltwater is less dense relative to the salty surroundings, so it tends to linger near the ice and insulate it from warmer water, slowing the melt. To make the flow visible, colored ice and dye are used to track the currents, showing strong downward streaming in fresh water and much weaker circulation in the saltwater case.
The third problem looks like a trick until the mechanics are understood: a metal ring dropped onto a closed loop of chain usually falls off, yet it can be made to “lock” onto the chain. The key is release timing and rotation. Letting the ring go from one side first—rather than dropping it symmetrically—introduces about a quarter-turn of rotation (~90 degrees). As the ring slides down, parts of it ride up the chain links and then, near the bottom, the geometry and motion cause the ring to snap into a position where it becomes trapped, effectively “sucked” into the middle and then pulled around to lock.
Taken together, the problems emphasize that everyday surprises often come down to equilibrium versus non-equilibrium behavior, density-driven fluid motion, and how small changes in initial conditions can redirect motion. Shaking a soda bottle, adding salt to water, or releasing a ring a fraction differently each changes the microscopic pathways—nucleation sites, convection currents, or rotational alignment—that determine what happens next.
Cornell Notes
Shaken carbonated drinks can overflow without any real increase in headspace pressure. After a bottle sits, dissolved CO2 and gas in the headspace are in equilibrium at about 3 atmospheres; shaking mainly adds nucleation sites (tiny bubbles) that let CO2 come out of solution rapidly once pressure drops on opening. In the ice experiment, fresh water melts faster because cold meltwater is denser than the surrounding liquid, driving convection that brings warm water to the ice. In saltwater, the cold meltwater is relatively less dense, so it stays near the ice and insulates it, slowing melting. A ring can also lock onto a closed chain loop when released in a way that induces rotation, letting the ring snap into a trapped geometry.
Why doesn’t shaking a carbonated bottle increase the pressure in the headspace?
What actually makes a shaken soda overflow more easily?
How does saltwater slow ice melting compared with fresh water?
How can a ring lock onto a closed loop of chain even though it normally falls off?
How can nucleation sites be added without shaking?
Review Questions
- Explain the difference between equilibrium and non-equilibrium conditions in the carbonated-drink scenario, and how that difference changes CO2 release rates.
- Use density and convection to predict which environment (fresh water or saltwater) should produce faster ice melting and why.
- Describe how rotation and release asymmetry enable a ring to become trapped on a closed chain loop.
Key Points
- 1
Shaking a carbonated drink doesn’t meaningfully raise headspace pressure; it mainly adds nucleation sites (tiny bubbles) in the liquid.
- 2
A sealed soda bottle sitting at room temperature reaches equilibrium where dissolved CO2 matches the CO2 pressure in the headspace (about 3 atmospheres).
- 3
Opening the bottle drops pressure to ambient, letting CO2 come out of solution rapidly when nucleation sites are present, causing overflow.
- 4
Fresh water melts ice faster because cold meltwater is denser and sinks, driving convection that brings warmer water to the ice.
- 5
Saltwater slows melting because cold meltwater is relatively less dense than the surrounding salty solution, reducing convection and insulating the ice.
- 6
A ring can lock onto a closed chain loop when released asymmetrically so rotation (~90 degrees) and sliding geometry cause it to snap into a trapped configuration.
- 7
Rough surfaces like Mentos can create nucleation sites and trigger faster CO2 release without shaking.