Alzheimer's and the Brain
Based on Vsauce's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Alzheimer’s disease involves progressive cognitive decline tied to brain cell damage, not normal aging.
Briefing
Alzheimer’s disease is driven by physical damage inside the brain—especially the buildup of sticky protein structures—yet it also remains stubbornly hard to predict or stop. The condition affects memory and thinking, and it becomes more common with age, but it isn’t considered a normal part of aging. From 2000 to 2013, Alzheimer’s rose as a cause of death even as many other causes declined, underscoring why researchers keep searching for ways to prevent it, delay it, or slow its progression.
At the center of the disease story is how the brain stores information. Learning doesn’t simply add new “storage space” or create new wrinkles in the brain’s surface; instead, memories are thought to form through strengthened connections between neurons. When neurons repeatedly fire together, the connections become more likely to activate again—making recall easier. That framework helps explain why memory fades when brain cells or their connections are damaged, blocked, compromised, or killed, and why forming new memories can become harder.
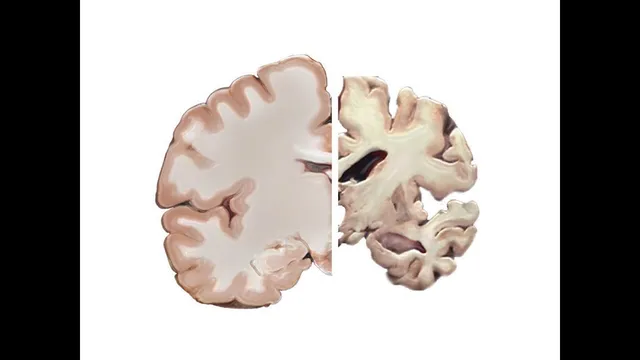
Alzheimer’s is linked to two major microscopic abnormalities: plaques and tangles. Plaques form when fragments of a protein called amyloid precursor protein (APP) are processed in ways that produce amyloid-beta. Enzymes such as alpha-secretase and beta-secretase cut APP into different fragments, and gamma-secretase can further process them. When amyloid-beta isn’t cleared efficiently—or when it folds incorrectly—small clumps called oligomers can form. Those oligomers can then grow into larger structures known as plaques. Plaques are associated with brain cell damage, and oligomers are suspected to be especially toxic, potentially allowing unregulated ion flow into neurons.
Tangles involve another protein, tau (often referred to as “TOA” in the transcript), which normally stabilizes microtubules—cellular “railroad ties” that help guide nutrients and other materials. In Alzheimer’s, tau becomes abnormally sticky and tangles with other tau proteins, disrupting the delivery network inside neurons.
Genetics offers clues but not a complete answer. Only about 1–5% of cases are linked to identifiable genetic differences, while most causes remain unclear. Still, strong evidence comes from Down syndrome: roughly 15% of people with Down syndrome who live to 40 or longer develop Alzheimer’s, and among those who reach 60, estimates rise to 50–70%. Down syndrome includes an extra copy (or part) of chromosome 21, which contains instructions for APP—connecting chromosome 21 directly to amyloid-related pathology.
Risk reduction strategies are also part of the practical picture. Factors associated with lower risk include physical activity, a heart-healthy diet, learning a second language later in life, staying socially engaged, protecting the head from injury, and ongoing intellectual activities such as crossword puzzles, playing instruments, reading, and board games. The goal isn’t just to treat Alzheimer’s after it appears, but to build cognitive resilience—keeping the brain’s networks healthier for longer so that, if damage occurs, it may be delayed or better tolerated.
Cornell Notes
Alzheimer’s disease is marked by progressive cognitive decline tied to physical changes in the brain, especially amyloid plaques and tau tangles. Memory formation is described as strengthening connections between neurons through repeated firing, so damage to cells and their links can erase or weaken recall and learning. Plaques are linked to how amyloid precursor protein (APP) is cut by enzymes (alpha-secretase, beta-secretase, gamma-secretase), producing amyloid-beta fragments that can clump into oligomers and then plaques. Tau normally stabilizes microtubules, but in Alzheimer’s it becomes abnormally sticky, disrupting intracellular transport. Genetics explains a small fraction of cases, while risk-reduction behaviors—exercise, diet, social and intellectual engagement, and head protection—are associated with lower risk and may improve brain resilience.
How does the transcript connect learning and memory to brain structure and neuron activity?
What are plaques, and how do they form from APP in the transcript’s explanation?
Why are oligomers and plaques treated differently in terms of toxicity?
What role does tau play normally, and what goes wrong in Alzheimer’s?
How does Down syndrome strengthen the genetic link to Alzheimer’s in the transcript?
Which lifestyle factors are associated with reduced Alzheimer’s risk, and what’s the underlying goal?
Review Questions
- What connection-based mechanism is proposed for how memories form, and how does Alzheimer’s disrupt it?
- Trace the APP processing pathway described (alpha-secretase, beta-secretase, gamma-secretase) and explain how it leads to oligomers and plaques.
- How do plaques and tangles differ in what proteins they involve and what cellular functions they disrupt?
Key Points
- 1
Alzheimer’s disease involves progressive cognitive decline tied to brain cell damage, not normal aging.
- 2
Memory formation is framed as strengthened neuron-to-neuron connections created by repeated firing, so damaged connections can erase or weaken recall.
- 3
Amyloid plaques are linked to APP processing into amyloid-beta fragments; misprocessing, slow clearance, or misfolding can produce oligomers that grow into plaques.
- 4
Tau tangles disrupt microtubule stability and interfere with intracellular transport, undermining neuron function.
- 5
Only a small fraction of Alzheimer’s cases have identifiable genetic differences, but Down syndrome strongly implicates chromosome 21 and APP.
- 6
Risk is associated with modifiable factors such as exercise, heart-healthy diet, social engagement, intellectual activity, head protection, and learning later in life.