Cruel Bombs
Based on Vsauce's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Nuclear detonations produce intense heat and radiation, including radioactive isotopes such as Strontium-90 and Caesium-137.
Briefing
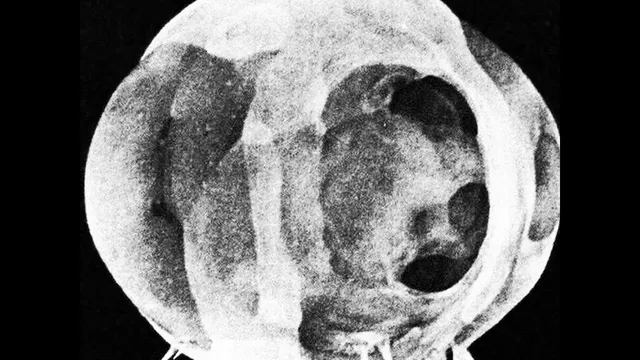
Nuclear weapons are built to unleash temperatures and radiation that can gut atoms and vaporize matter in fractions of a second—but the real story is how thin the margin is between deterrence and catastrophe. Detonation produces a “lining” of radioactive isotopes such as Strontium-90 and Caesium-137, and the energy release is so intense that atoms are effectively ripped apart at temperatures exceeding the surface of the Sun. High-speed photography from Harold Edgerton’s rapatronic camera captured nuclear fireballs less than a thousandth of a second after detonation, using a magnetic shutter to freeze the event at a billionth-of-a-second exposure—showing how the blast energy can turn supporting metal into plasma.
The transcript then pivots from physics to risk: even a small fraction of nuclear material can be devastating. When “Little Boy” was detonated over Hiroshima, only 1.38% of its uranium actually fissioned; the rest was blown away before it could react. Yet the fission of just 0.7 grams of uranium—described as less than the weight of a banknote—was enough to kill about 80,000 people and destroy roughly two-thirds of the city’s buildings. With tens of thousands of nuclear weapons worldwide, accidents become a persistent possibility: fires, miscommunication, rogue actions, or a dropped warhead.
A key quantitative comparison frames how unlikely but not impossible nuclear mishaps are. The acceptable probability of a nuclear weapon accident is given as one in a million. For context, in 2012 the odds of dying in a commercial airliner accident were about one in forty million—meaning nuclear accidents are treated as more likely than that benchmark, even though both are rare.
Concrete “oops” moments underline the stakes. In North Carolina, a US B-52 bomber carrying two 4 megaton thermonuclear bombs tumbled from the sky when a loose lanyard snagged the bomb release switch. Lieutenant Jack ReVell later noted that only one safety mechanism—a low-voltage arming switch—didn’t fail, preventing a nuclear catastrophe. One bomb’s uranium-rich secondary was never recovered and remains buried underground.
The transcript also broadens the theme of human error beyond state arsenals. A 17-year-old, David Han, attempted to build a nuclear reactor in his mother’s backyard in Michigan in 1994, drawing on radioactive materials already present in everyday items like smoke detectors (Americium) and glow-in-the-dark paint (Radium). His reactor never reached critical mass, but it reportedly exposed his neighborhood to about 1,000 times normal background radiation, leading to a Superfund cleanup and confiscation of his work. Later, David Hahn was arrested for stealing smoke detectors, and his sores were believed to be linked to radioactive exposure.
World War II provides additional lessons about uncertainty. Charles Sweeney flew a B-29 over Kokura with the bomb bay doors open for nearly an hour, but cloud cover prevented visual confirmation, forcing a switch to Nagasaki—where about 75,000 people died. The transcript closes by contrasting humanity’s ability to build “cruel bombs” with its limits in controlling outcomes, echoing Richard Feynman’s view that science supplies keys to power but doesn’t dictate whether they open heaven or hell.
Cornell Notes
Nuclear weapons combine extreme heat and radiation with a sobering reality: only a tiny fraction of nuclear material may need to fission to cause mass destruction. “Little Boy” over Hiroshima used just 1.38% of its uranium, yet about 0.7 grams of fissioned material was enough to kill roughly 80,000 people and destroy two-thirds of the city. Because large arsenals increase the chance of accidents, the transcript cites an “acceptable” nuclear weapon accident probability of one in a million—still rare, but not negligible. Real incidents, such as a B-52 crash in North Carolina where a low-voltage arming switch prevented detonation, show how safety design can matter. It also highlights how uncertainty—like wartime cloud cover—can redirect catastrophic outcomes.
Why does the transcript emphasize that only a small fraction of uranium fissioned in Hiroshima?
What does Edgerton’s rapatronic camera footage add to the discussion beyond numbers?
How is nuclear accident risk quantified, and how does it compare to airline risk?
What prevented a nuclear catastrophe in the North Carolina B-52 incident?
How does the transcript connect everyday materials to nuclear risk through David Han’s reactor attempt?
Why does wartime targeting uncertainty matter in the transcript’s broader theme?
Review Questions
- What does the Hiroshima “1.38% fission” detail imply about how nuclear damage scales with fuel utilization?
- Which safety mechanism is credited with preventing detonation in the North Carolina B-52 crash, and what happened to part of one bomb afterward?
- How do cloud cover and limited weather prediction illustrate uncertainty in catastrophic decision-making?
Key Points
- 1
Nuclear detonations produce intense heat and radiation, including radioactive isotopes such as Strontium-90 and Caesium-137.
- 2
Only a small fraction of fissile material can be enough for mass destruction; “Little Boy” fissioned 1.38% of its uranium yet caused catastrophic damage.
- 3
Risk management for nuclear weapons uses probabilistic benchmarks, including an “acceptable” accident probability of one in a million.
- 4
Real-world incidents show how safety mechanisms can prevent disaster, such as the low-voltage arming switch in a B-52 crash in North Carolina.
- 5
Uncertainty in targeting and environment—like wartime cloud cover—can change which city is struck even when a primary target is selected.
- 6
Radioactive materials are not limited to weapons; everyday items like smoke detectors and glow-in-the-dark paint can contain Americium and Radium.
- 7
Science can identify powerful “keys,” but it doesn’t determine whether they’re used responsibly—human choices decide outcomes.