How Can Matter Be BOTH Liquid AND Gas?
Based on PBS Space Time's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
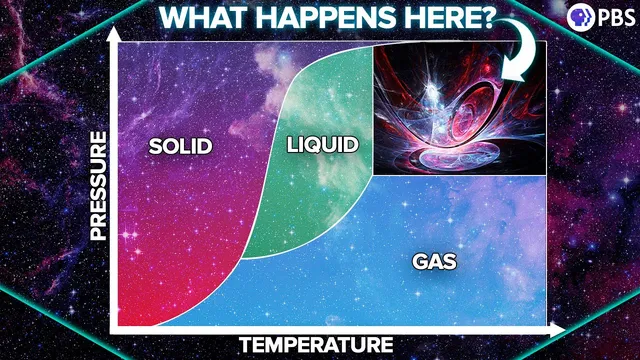
The liquid–gas boundary ends at the critical point; beyond it, matter becomes a supercritical fluid with no distinct boiling/condensing transition.
Briefing
Supercritical fluids let matter behave like a hybrid of liquid and gas—crossing the liquid–gas “no-man’s land” at the critical point—unlocking properties that neither ordinary liquids nor gases can match. The key insight is that when temperature and pressure rise together past the end of the liquid–gas boundary, the distinction between “boiling” and “condensing” disappears. Instead of a sharp phase change, the fluid can continuously shift from liquid-like density to gas-like flow, producing a state that is compressible like a gas but has liquid-like density, with no surface tension.
That behavior is rooted in how liquids and gases respond to pressure and temperature. Liquids resist compression, maintain a distinct surface tension, and expand with heat; gases are easy to compress, fill their container, and exert pressure through particle impacts. On a phase diagram, changing temperature and pressure moves a substance across boundaries where it becomes a momentary mixture of phases. But the liquid–gas boundary doesn’t extend forever: it ends at the critical point. Beyond that point lies the supercritical region where matter can “skirt around” the phase boundary—so it can become effectively gas-like without ever undergoing a conventional boiling transition.
Carbon dioxide provides a concrete path into the supercritical state. Starting from dry ice (solid CO2), warming at atmospheric pressure triggers sublimation into gas, raising pressure inside a sealed chamber. Heating further increases both temperature and pressure, and the rising pressure also lifts CO2’s boiling point, creating a feedback loop that suppresses evaporation. As the system approaches the critical point, the densities of the liquid and gas converge: gas density rises as it compresses, while liquid density falls as it thermally expands. Once those densities match, microscopic liquid droplets can diffuse through the gas, and the entire sample becomes supercritical CO2. Visually it can look like a gas—often transparent—yet it behaves differently under the hood.
Supercritical CO2 combines gas-like transport with liquid-like intermolecular interactions. It flows and diffuses with very low viscosity, lacks liquid surface tension, and is compressible like a gas. But its high density means particles interact strongly, so it follows a more complex equation of state than the ideal gas law. Experiments with silica beads illustrate the hybrid character: in the supercritical state the beads move only slightly, resembling liquid-like damping, whereas in a purely gaseous environment they rattle freely.
The practical payoff is that supercritical fluids can dissolve substances like a solvent while still moving through materials like a gas. That “super-solvent” capability underpins decaffeination: supercritical CO2 diffuses into coffee beans, binds caffeine, and then is converted back to gas so caffeine can be recovered. It also enables aerogel production by replacing water in a fragile gel scaffold, preserving a low-density lattice full of air. Beyond consumer uses, supercritical fluids matter in materials science, heat transfer, power plants, heat pumps, and refrigeration—using supercritical carbon dioxide, hydrocarbons, or water depending on the chemistry and engineering needs.
Nature also hosts supercritical fluids. Deep geothermal water can reach the needed temperature and pressure near hydrothermal vents. Venus is described as an “ocean world” of supercritical CO2 because its greenhouse-heated surface and extreme pressure place CO2 near or beyond the supercritical point. Even the gas giants are framed as having supercritical layers—hydrogen or other fluids—beneath their thick gaseous atmospheres.
The episode closes by tying the idea to a broader theme: while many exotic states are rare or extreme, supercritical fluids are increasingly useful and surprisingly common across Earth and space, even if they remain invisible to everyday intuition.
Cornell Notes
Matter’s liquid–gas distinction breaks down at the critical point. When temperature and pressure rise together past the end of the liquid–gas boundary, there’s no sharp boiling transition—matter becomes a supercritical fluid that is compressible like a gas but dense like a liquid, with no surface tension. Supercritical CO2 is used as the main example: heating dry ice in a sealed chamber raises pressure and boiling point until the liquid and gas densities converge, producing a transparent, gas-like fluid with liquid-like interactions. Because it dissolves like a solvent yet flows like a gas, supercritical fluids enable processes such as decaffeination, aerogel production, and industrial materials deposition. They also occur naturally in hydrothermal systems and in Venus’s high-pressure CO2 environment.
What exactly is the “critical point,” and why does it matter for phase changes?
How do pressure and temperature jointly determine whether CO2 is solid, liquid, gas, or supercritical?
Why does supercritical CO2 look like a gas but behave differently?
What experimental signposts show the “hybrid” nature of supercritical fluids?
Why are supercritical fluids such effective solvents for processes like decaffeination and aerogel drying?
Where does supercritical matter occur naturally, beyond lab setups?
Review Questions
- How does the phase diagram explain why supercritical fluids can transition from liquid-like to gas-like behavior without a sharp boiling point?
- In the CO2 chamber example, what role does pressure play in raising the boiling point and preventing evaporation during heating?
- What combination of properties makes supercritical CO2 useful as both a solvent and a transport medium for diffusion-based industrial processes?
Key Points
- 1
The liquid–gas boundary ends at the critical point; beyond it, matter becomes a supercritical fluid with no distinct boiling/condensing transition.
- 2
Supercritical fluids are compressible like gases but have liquid-like density and no surface tension.
- 3
High density in the supercritical regime increases particle interactions, so behavior cannot be captured by the ideal gas law alone.
- 4
Supercritical CO2 can be produced by heating dry ice in a sealed chamber until temperature and pressure drive the system past the critical point.
- 5
Supercritical fluids act as “super-solvents” because they dissolve like dense liquids while diffusing and flowing like gases.
- 6
Practical uses include decaffeination, aerogel production, and industrial processing such as materials deposition and heat-transfer applications.
- 7
Supercritical fluids occur naturally in hydrothermal environments on Earth and in Venus’s high-pressure CO2 atmosphere, and may form layers inside gas giants.