Reimagining the Periodic Table
Based on minutephysics's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
The standard periodic table’s left-right breaks (including the transition around elements 10 and 11) are treated as drawing conventions rather than physical necessities.
Briefing
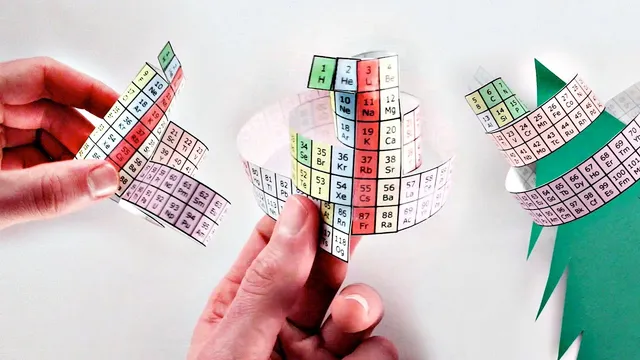
The periodic table’s most familiar layout is more a convention than a necessity: its “breaks” between certain elements are artificial, and the underlying pattern can be made continuous by rearranging the table into loops, spirals, and folds. Instead of treating the left and right edges as permanently separated, the transcript argues that the table should be cut and taped into a cycle—much like how map boundaries can suggest a discontinuity that doesn’t exist in physical geography.
In the standard periodic table, the transition from element 10 to 11 appears as a glaring structural jump, with a tall column on the left, a gap, and then tall columns on the right. The proposed fix is to remove those artificial discontinuities and connect the edges, producing a “left step” periodic table where columns descend in a stair-step pattern. A further refinement shifts helium upward so it sits above beryllium, and places hydrogen where it “obviously belongs,” yielding a layout that aligns more naturally with physics-based expectations—even though it sacrifices some of the classic visual trends (like electronegativity and first ionization energy increasing left-to-right and bottom-to-top).
But the loop idea runs into a new problem: even after connecting the edges, there can still be a gap between specific element ranges (notably between elements 20 and 21). The transcript then explores alternative connections—pairing elements 4 and 5, for example—to eliminate gaps entirely. That produces a “spiral periodic table,” a continuous arrangement with no missing numbers. The tradeoff is structural: elements with similar properties no longer stack neatly in vertical columns, so the periodic “grouping” becomes harder to read.
To restore vertical grouping, the spiral can be “folded” so that elements that should share a column end up aligned again. This leads to a more complex, three-dimensional-looking form described as a “spiral rosette” (also nicknamed “Mendeleev’s flour”). It highlights the table’s underlying structure more clearly than the flat versions, but it stops being a practical two-dimensional reference.
The final step is a return to basics: the one-dimensional periodic table. Although it’s long and less visually intuitive, it preserves the core fact that properties repeat periodically. By matching repeated patterns and performing careful “surgery” (cutting and rearranging at the right points), the familiar traditional periodic table can be reconstructed—suggesting that the classic layout is a convenient projection of deeper periodic structure rather than the only meaningful way to organize elements.
Overall, the transcript treats the periodic table as a flexible representation of repeating physics, showing how different geometries—loop, spiral, rosette, and line—trade readability for structural continuity.
Cornell Notes
The periodic table’s standard layout contains artificial breaks that can be removed by reconnecting the ends, turning the table into a loop. Recutting and rejoining elements yields new geometries: a stair-stepped “left step” table, then a gap-free “spiral” when different edge connections are chosen (like pairing elements 4 and 5). A spiral eliminates numeric gaps but disrupts vertical grouping of similar properties, so restoring those groups requires folding into a “spiral rosette” (Mendeleev’s flour), which becomes too three-dimensional for everyday use. The exercise ends by returning to a one-dimensional, repeating-pattern view—then reconstructing the familiar table by cutting at the right places.
Why are the breaks in the traditional periodic table considered “artificial,” and what does looping change?
What is the “left step” periodic table, and what physics-based adjustment improves it?
How does the spiral periodic table eliminate gaps, and what tradeoff does it introduce?
What forces the spiral to become a folded “rosette,” and why is it impractical?
Why does the transcript end with a one-dimensional periodic table, and how does it recover the familiar layout?
Review Questions
- What specific discontinuity in the standard periodic table motivates the “cut and tape into a loop” idea?
- How do the spiral and rosette arrangements differ in their ability to preserve vertical grouping of similar properties?
- What method does the transcript use to move from a one-dimensional repeating-pattern view back to the traditional periodic table?
Key Points
- 1
The standard periodic table’s left-right breaks (including the transition around elements 10 and 11) are treated as drawing conventions rather than physical necessities.
- 2
Connecting the table’s ends turns it into a loop, removing the apparent discontinuity created by the conventional layout.
- 3
A “left step” arrangement can look more natural, especially when helium is moved above beryllium and hydrogen is placed in its preferred position.
- 4
Eliminating numeric gaps entirely may require different edge connections, such as joining elements 4 and 5 to produce a gap-free spiral.
- 5
Spiral layouts preserve continuity but disrupt vertical grouping, so restoring group alignment requires folding into a rosette.
- 6
A one-dimensional periodic view emphasizes repeating patterns; careful cutting and rearranging can reconstruct the familiar table from that underlying repetition.