Something Strange Happens When You Trust Quantum Mechanics
Based on Veritasium's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
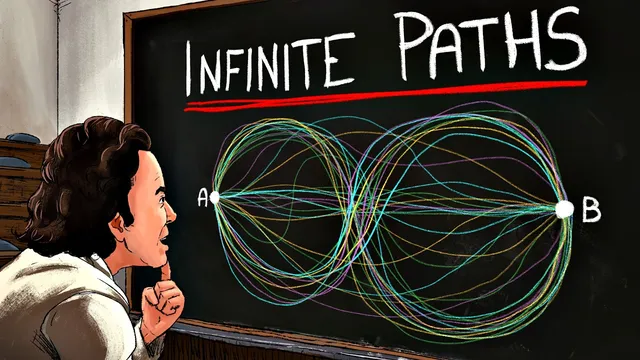
Quantum particles are treated as contributing amplitudes from every possible path between two points, not just one trajectory.
Briefing
Quantum particles don’t follow a single, definite route between two points. Instead, they effectively “try” every possible path at once, and the paths that survive are the ones whose contributions add up coherently. The reason the world still looks classical—balls arc predictably, light reflects at a single angle, planets keep stable orbits—is that almost all alternative routes cancel out through destructive interference. What matters is a quantity called action: it controls the phase of a particle’s wave-like contributions, so only paths near the least-action route reinforce each other.
The argument starts with a familiar optimization problem: when someone can run on land and swim in water, the fastest route from point A to point B isn’t purely straight or purely along the shoreline. The optimal path depends on the relative speeds, and the same math governs how light bends when it enters a new medium. That raises a puzzle: light seems to “know” the route that minimizes travel time, yet classical intuition says light should just propagate locally—changing direction only when it hits a boundary.
The bridge to quantum mechanics is the principle of least action. Historically, action was introduced as a way to reformulate mechanics: instead of tracking forces directly, physics can be computed by finding the path that minimizes action. Hamilton later connected action to an integral of kinetic energy minus potential energy. The transcript then traces how this idea reappears at the birth of quantum theory, beginning with blackbody radiation. Classical physics predicted the “ultraviolet catastrophe,” an infinite energy output at short wavelengths, because it allowed atoms to emit electromagnetic waves with arbitrarily small energies. Max Planck resolved the mismatch by quantizing energy: electromagnetic energy comes in discrete packets, E = hf, where h is Planck’s constant. Crucially, h has units of action, hinting that nature’s fundamental bookkeeping is about action quanta, not just energy.
Einstein used Planck’s quantization to explain the photoelectric effect, and Bohr applied quantization to atomic stability by discretizing angular momentum in units of ħ. De Broglie then provided the missing physical intuition: if matter has wave properties, electrons in atoms must form standing waves. That wave requirement forces quantized orbits and reproduces hydrogen’s spectral lines.
From there, the transcript returns to the double-slit logic and generalizes it: if it’s impossible to assign a single slit, the particle’s wave must include contributions from both slits. Extending that reasoning to many slits leads to the conclusion that every path contributes. Feynman’s formulation makes this concrete: each path carries a complex amplitude whose phase depends on the action accumulated along that route. Because ħ is tiny, the phases of most paths swing wildly and cancel out, leaving only paths near the least-action trajectory.
A hands-on demonstration is used to make the interference claim tangible. By covering a mirror in fine strips, the usual reflection pattern breaks into multiple spots—evidence that different path contributions can be selectively un-canceled. A follow-up laser version reinforces the point: when the “canceled” regions are restored in the right pattern, the missing reflections reappear. The closing takeaway is that modern physics is organized around Lagrangians and action, and the ongoing search for a “theory of everything” is, in essence, the hunt for the correct underlying mathematical structure that yields all laws from one action principle.
Cornell Notes
The transcript argues that quantum objects don’t take one definite trajectory. Instead, they contribute amplitudes from every possible path between two points, and the observed outcome comes from interference. The phase of each path’s contribution is tied to the action accumulated along that route; because Planck’s constant ħ is extremely small, phases for most paths vary so rapidly that they cancel out. Paths near the least-action route add up coherently, which is why classical mechanics emerges as an approximation. The blackbody radiation problem, Planck’s quantization (E = hf), and later Einstein, Bohr, and de Broglie developments are used to show how action and quantization become central to quantum theory.
Why does the “fastest path” idea for light matter for the quantum claim about paths?
How does Planck’s solution to blackbody radiation connect to action rather than just energy?
What role do Einstein, Bohr, and de Broglie play in turning quantization into a physical mechanism?
How does the double-slit reasoning generalize into “all possible paths” contributing?
What determines which paths survive in Feynman’s formulation?
How do the mirror/foil and laser demos support the interference-and-paths claim?
Review Questions
- What is the relationship between action and the phase of a quantum path in the transcript’s Feynman-style explanation?
- Why do most alternative paths cancel out even though quantum mechanics requires summing over them?
- How do Planck’s quantization and the units of Planck’s constant (action) set up the later least-action/phase argument?
Key Points
- 1
Quantum particles are treated as contributing amplitudes from every possible path between two points, not just one trajectory.
- 2
The observed “single outcome” arises because amplitudes interfere: most paths cancel through destructive interference.
- 3
Action determines the phase accumulation along a path; the transcript links this to an integral of kinetic energy minus potential energy.
- 4
Planck’s constant h (and ħ) carries units of action, tying early quantum breakthroughs to the later action-based phase picture.
- 5
Blackbody radiation’s “ultraviolet catastrophe” is resolved by energy quantization E = hf, which prevents infinite high-frequency emission.
- 6
Einstein, Bohr, and de Broglie build a chain from quantized light to quantized atomic states via wave behavior and standing-wave conditions.
- 7
Fine-pattern mirror and laser demonstrations are used to show that changing which path contributions add coherently shifts where reflection appears.