Why Super Glue Is Perfect For Gluing Skin
Based on Veritasium's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Super glue’s rapid set comes from ethyl cyanoacrylate monomers polymerizing via a water-triggered chain reaction, typically within 10–30 seconds.
Briefing
Super glue’s strength comes from fast chemistry: ethyl cyanoacrylate monomers spread into tiny pores and crevices, then rapidly polymerize when they encounter water (from the air, surfaces, or skin). That combination—mechanical “keying” into microscopic texture plus a chain-reaction that turns liquid into rigid polymer—explains why a single drop can hold enormous loads, why it sets in 10–30 seconds, and why it can be so difficult to remove from living tissue.
The adhesive story begins in World War II-era materials research. Chemist Harry Coover worked on cyanoacrylate while Eastman Kodak sought a clear, castable plastic to replace glass lenses. The compound’s defining flaw—sticking to almost everything—was initially a “severe pain.” A coworker ruined an expensive refractometer by measuring it the wrong way, but Coover reframed the problem. He started gluing together whatever he could reach—glass, rubber, metal, wood, paper—and found bonds that formed almost instantly and resisted breaking. The company later branded it Eastman 910 Adhesive, and the world eventually knew it as super glue.
At the molecular level, super glue in a tube is a liquid of ethyl cyanoacrylate monomers. When applied between two surfaces, the liquid flows into microscopic gaps. Polymerization begins because ethyl cyanoacrylate is chemically “primed” to react: electron-deficient bonding sites (involving a nitrile group and an ester group) are hungry for electrons. Water supplies the trigger. Slightly negative oxygen atoms in water and negative hydroxide ions initiate the reaction by breaking open the reactive carbon double bond, starting a chain reaction that links monomers into long polymer chains. Each water molecule can initiate new chains, which is why super glue sets quickly on most surfaces that carry moisture—air humidity, surface water, or water absorbed into materials like fabrics.
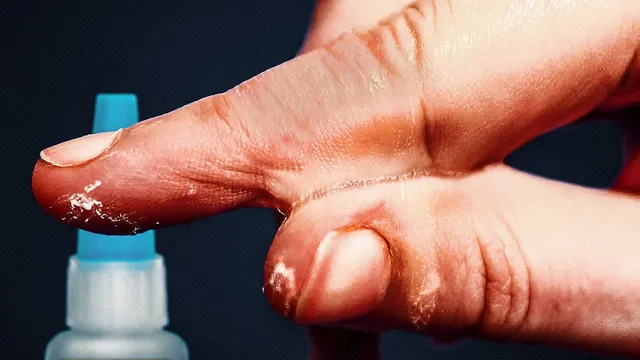
Skin is an especially effective substrate. Wrinkles and pores provide physical anchoring, and collagen’s chemistry offers additional negative regions that can help initiate polymerization. That same mechanism underlies forensic uses: super glue fumes can lift fingerprints from non-porous surfaces because handling leaves moisture and oils.
Strength has limits, though. Super glue forms dense, mostly linear polymer chains that make it rigid and strong in compression and tension along the chain direction, with tensile strength reported up to about 25 MPa. But it is brittle. Sudden impacts, shear forces (especially perpendicular to chain direction), and peeling concentrate stress at edges, causing fractures or “zipper-like” unzipping. It also fails completely on certain plastics—polyethylene, polypropylene, and Teflon—because their surfaces are chemically inert and hydrophobic, offering neither reactive sites nor water-based initiation.
Medical and environmental implications followed. Coover pursued tissue-safe versions after discovering that pure cyanoacrylate releases heat as it polymerizes, can degrade into toxic byproducts like formaldehyde, and is too brittle for flexible healing. Increasing the alkyl chain length slowed reaction rate, reduced heat spikes, delayed toxic breakdown, and improved flexibility. The resulting product, approved in 1998 as Dermabond (2-octyl cyanoacrylate), became a major industry.
Finally, researchers are exploring super glue’s unusual recyclability. Unlike typical plastics that degrade when mechanically or thermally recycled, cyanoacrylate can be depolymerized: heating to about 210°C breaks it back into monomers that can be distilled and reactivated into fresh polymer. The challenge is engineering a plastic that can be handled and cast while avoiding super glue’s brittleness—an obstacle that modern chemistry aims to solve by controlling initiators, chain length, and polymer flexibility.
Cornell Notes
Super glue works because ethyl cyanoacrylate monomers spread into tiny pores and crevices, then rapidly polymerize into rigid chains when triggered by water. The chemistry is electron-deficient and “hungry” for electrons, so water (and hydroxide ions) initiates a chain reaction that turns a liquid into a solid in about 10–30 seconds. That same mechanism makes skin an ideal bonding surface and explains fingerprint-lifting with super glue fumes. Strength is high in compression and tension along polymer chains, but brittleness makes it fail under impact, shear, and peeling. It won’t bond to chemically inert, hydrophobic plastics like polyethylene, polypropylene, and Teflon, which lack reactive sites and don’t hold water.
Why does super glue set so fast—what actually triggers polymerization?
How does super glue achieve both strong bonding and fast setting on many surfaces?
Why is super glue strong in some directions but weak in others?
Why doesn’t super glue stick to polyethylene, polypropylene, or Teflon?
What made cyanoacrylate suitable for medical use, and why did the original form fail?
How could super glue help with plastic recycling?
Review Questions
- What roles do water and surface texture play in super glue’s rapid setting and strong adhesion?
- Describe two mechanical loading modes where super glue performs poorly and explain why brittleness and chain direction matter.
- Why do chemically inert, hydrophobic plastics prevent super glue from bonding even when water is present?
Key Points
- 1
Super glue’s rapid set comes from ethyl cyanoacrylate monomers polymerizing via a water-triggered chain reaction, typically within 10–30 seconds.
- 2
Strong adhesion relies on both chemical polymer formation and physical anchoring into pores and crevices; smooth surfaces bond poorly unless roughened.
- 3
Super glue is directionally strong (compression and tension along polymer chains) but brittle, making it vulnerable to impact, shear, and peeling.
- 4
Super glue fails on polyethylene, polypropylene, and Teflon because their surfaces are chemically inert and hydrophobic, preventing both electron donation and water-based initiation.
- 5
Medical cyanoacrylate adhesives like Dermabond work by slowing reaction rate through longer alkyl chains, reducing heat spikes and delaying toxic breakdown.
- 6
Super glue’s depolymerization potential (around 210°C) offers a route to recycling by converting polymer back into monomers for re-polymerization.