S8E2 - Electronegativity, Dipole Moments, and Bond Polarity
Based on ChemistryNotes Videos's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Electronegativity increases across the periodic table from bottom-left to top-right, making top-right elements like fluorine strong electron attractors.
Briefing
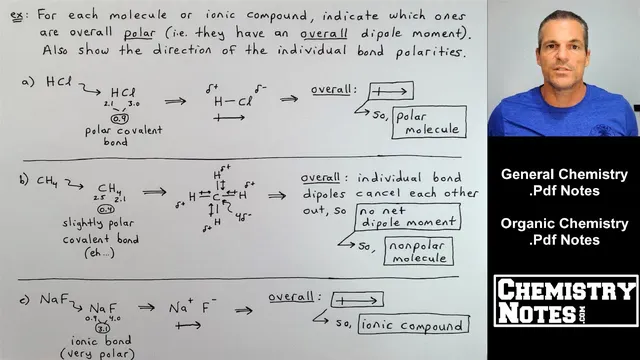
Whether a bond is ionic or covalent—and whether it makes a molecule polar—comes down to electronegativity differences between the bonded atoms. Electronegativity values run roughly from 0.7 to 4.0 across the periodic table, increasing from bottom-left to top-right. Fluorine and chlorine sit at the high end and strongly “pull” shared electrons toward themselves, like a tug-of-war where the more electronegative atom wins.
To classify bond type, the method is straightforward: subtract the electronegativity values of the two atoms and compare the absolute difference to a polarity chart. A difference of 3.3 is the maximum and corresponds to about 100% ionic character. Between about 0.3 and 1.7 falls the polar covalent range, while below 0.3 is nonpolar covalent. Using this approach, HH has an electronegativity difference of 0 and is nonpolar covalent. OH is 1.4 (polar covalent), SH is 0.4 (polar covalent), and FH is 1.9 (ionic by the chart’s cutoff). NaCl has a difference of 2.1, placing it firmly in the ionic category and making it the most polar among the listed bonds.
Once bond polarity is known, dipole moment provides the direction of that polarity. A dipole moment is drawn as an arrow pointing toward the more electronegative atom, with partial charges indicated by δ+ and δ−. In HCl, the shared electron pair is pulled toward chlorine, producing partial negative charge on Cl and partial positive charge on H; the electron density is uneven but not fully transferred. In NaCl, the electronegativity gap is large enough that the electron is effectively transferred, giving Na a full +1 and Cl a full −1, matching the ionic picture.
Those same ideas extend from individual bonds to whole molecules or ionic formula units. A molecule is overall polar only if the bond dipoles don’t cancel out. HCl is polar because its single bond dipole points toward Cl, giving a net dipole moment. CH4 is the classic cancellation case: four slightly polar C–H bonds point in different directions, and the geometry makes their dipole moments cancel, leaving no net dipole moment—so CH4 is nonpolar overall. NaF is treated as ionic and polar because the electron transfer produces full charges and a strong overall dipole.
For H2S, the S–H electronegativity difference is about 0.4, so each S–H bond is slightly polar covalent. With the correct molecular shape, the bond dipoles add rather than cancel, allowing an overall dipole moment and making H2S polar. The transcript emphasizes that drawing the molecule with the wrong orientation can falsely predict cancellation, a point reserved for later structure-drawing details.
Cornell Notes
Electronegativity differences determine whether a bond is ionic, polar covalent, or nonpolar covalent. Electronegativity increases from bottom-left to top-right on the periodic table, and the absolute difference between two atoms is compared to a chart: <0.3 nonpolar covalent, 0.3–1.7 polar covalent, and >1.7 ionic (up to ~3.3 for maximum ionic character). Bond polarity is represented with dipole moments, arrows pointing toward the more electronegative atom, using δ+ and δ− for partial charges. Overall molecular polarity depends on whether individual bond dipoles cancel out or add together. HCl and NaF are polar overall, CH4 is nonpolar due to cancellation, and H2S is polar when its bond dipoles don’t cancel.
How does electronegativity difference predict whether a bond is ionic, polar covalent, or nonpolar covalent?
Why does HCl have a dipole moment pointing toward Cl, and why is it only partial charges?
How does NaCl differ from HCl in dipole moment and charge description?
What determines whether a molecule is overall polar or nonpolar, even if it has polar bonds?
Using the transcript’s examples, which bonds are nonpolar covalent, polar covalent, and ionic?
Review Questions
- What electronegativity difference ranges correspond to nonpolar covalent, polar covalent, and ionic bonds on the chart?
- How do you draw a dipole moment for a single bond, and what do δ+ and δ− represent?
- Why is CH4 nonpolar overall even though each C–H bond has some polarity?
Key Points
- 1
Electronegativity increases across the periodic table from bottom-left to top-right, making top-right elements like fluorine strong electron attractors.
- 2
Bond type is determined by the absolute electronegativity difference: <0.3 nonpolar covalent, 0.3–1.7 polar covalent, and >1.7 ionic (with ~3.3 as the upper extreme).
- 3
Dipole moment indicates both the presence and direction of bond polarity, pointing toward the more electronegative atom.
- 4
Polar covalent bonds produce partial charges (δ+ and δ−) because electrons are shared unevenly rather than fully transferred.
- 5
Ionic bonds correspond to near-complete electron transfer, allowing full-charge descriptions like Na+1 and Cl−1.
- 6
Overall molecular polarity depends on whether bond dipole vectors cancel (CH4) or add (H2S).
- 7
Correct molecular orientation matters: an incorrect drawing can falsely predict dipole cancellation and the wrong overall polarity.