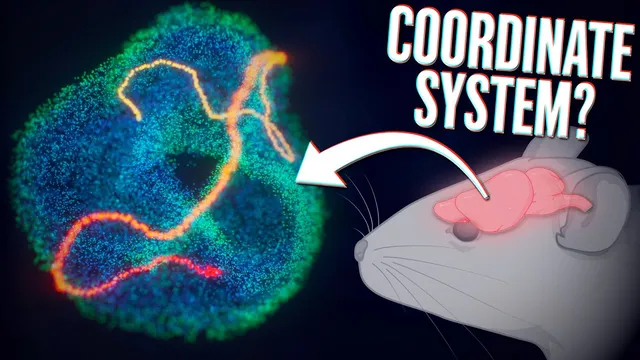
Your brain is moving along the surface of the torus 🤯
Based on Artem Kirsanov's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Grid cells encode spatial position using periodic hexagonal firing fields, but a single cell’s periodicity makes its activity ambiguous.
Briefing
Grid cells in the entorhinal cortex don’t just provide a “sense of direction” for navigation—they generate a low-dimensional geometric structure that behaves like a torus, letting the brain represent position in a way that stays stable across environments and even during sleep. The key finding is that when researchers analyze the collective firing patterns of many grid cells from the same module, the population activity forms a smooth, continuous trajectory constrained to a toroidal manifold. As an animal moves through space, its path in the real world corresponds to movement along this torus in neural state space, turning an apparently noisy neural code into a clean geometric signal.
The mechanism starts with how grid cells encode location. Each grid cell fires strongly at the vertices of a hexagonal tiling, producing “firing fields” that repeat periodically across the environment. A single cell is ambiguous: its activity could correspond to multiple possible positions spaced by the grid’s periodicity. The brain resolves that ambiguity by combining inputs from multiple grid-cell modules. Cells within a module share the same grid scale and orientation but have firing fields shifted relative to one another; different modules use different scales and orientations. Converging signals across modules allows the system to pinpoint a unique location.
To uncover the torus, the analysis treats the firing rates of many simultaneously recorded grid cells as coordinates in a high-dimensional space. Each moment in time yields a point determined by the instantaneous firing rates (derived from spike timestamps). When researchers apply dimensionality reduction to many such points, the high-dimensional cloud collapses onto a smooth three-dimensional embedding that reveals a toroidal structure. In this representation, each patch on the torus corresponds to a specific phase of the grid code—effectively a coordinate for position along the manifold.
The torus is notable not only for its geometry but for its invariances. The same toroidal population activity structure appears even when the animal is in different environments where individual grid-cell firing patterns look different to the eye. The network activity remains localized on the same torus, and the structure persists during sleep, when sensory input is absent. That combination points to an intrinsic origin: the toroidal manifold emerges from the network’s connectivity and synaptic weights rather than being imposed by incoming sensory information.
These results also connect to a broader theoretical framework: continuous attractor networks. In such models, constraints on network dynamics restrict activity to a subset of possible states, producing stable low-dimensional manifolds. The remaining open questions are how the torus arises from specific circuit architecture and whether other brain functions—potentially including higher-order cognition—use similar low-dimensional attractor geometries. For now, the central implication is clear: spatial navigation relies on a geometric, invariant neural substrate that turns complex population dynamics into a structured coordinate system.
Cornell Notes
Grid cells in the entorhinal cortex form a spatial coordinate system, but the new result is geometric: the collective firing of grid-cell populations from the same module traces a smooth trajectory on a torus in neural state space. A single grid cell’s periodic hexagonal firing is ambiguous, yet combining modules with different grid scales and orientations resolves position. Dimensionality reduction of multi-neuron firing rates reveals that population activity stays localized on the same toroidal manifold as the animal moves. Crucially, the torus persists across different environments and even during sleep, implying it is intrinsic to circuit connectivity and synaptic weights rather than driven by sensory input. This aligns with continuous attractor network theory and raises questions about other low-dimensional attractors in higher cognitive systems.
Why is activity from one grid cell not enough to determine position?
How do multiple grid-cell modules resolve that ambiguity?
What does it mean that population activity “maps to a torus”?
Why is the torus considered invariant across environments and during sleep?
How does this connect to continuous attractor network theory?
Review Questions
- What specific ambiguity arises from the periodic firing fields of a single grid cell, and how do module combinations remove it?
- Describe the analysis pipeline that turns multi-neuron spike data into evidence for a toroidal manifold.
- What experimental observations support the claim that the torus is intrinsic to circuit dynamics rather than imposed by sensory input?
Key Points
- 1
Grid cells encode spatial position using periodic hexagonal firing fields, but a single cell’s periodicity makes its activity ambiguous.
- 2
Modules of grid cells share scale and orientation within the module while differing across modules, enabling unique position decoding by combining signals.
- 3
Collective grid-cell firing rates, analyzed across many neurons, form a low-dimensional geometric structure with toroidal topology in neural state space.
- 4
As animals move, their neural population states trace a smooth trajectory along the torus, linking real-world movement to movement on the manifold.
- 5
The toroidal structure persists across different environments and during sleep, indicating it is intrinsic to network connectivity rather than driven by sensory input.
- 6
The findings align with continuous attractor network models, where connectivity restricts dynamics to stable low-dimensional manifolds.
- 7
Open questions remain about how the torus emerges from circuit architecture and whether similar attractor geometries support other cognitive functions.