Differential Equations: The Language of Change
Based on Artem Kirsanov's video on YouTube. If you like this content, support the original creators by watching, liking and subscribing to their content.
Neurons are modeled as dynamical systems whose present behavior depends on their evolving state over time, not just instantaneous input.
Briefing
Neurons behave less like simple input-output switches and more like time-dependent dynamical systems whose current response depends on their past. That temporal dependence is best understood through differential equations—the mathematical “language of change”—paired with geometric tools such as phase portraits and limit cycles, which reveal how complex behavior can emerge even from simple rules.
The foundation starts with dynamical systems theory: describe a system at any moment using state variables—real numbers that fully determine the system’s relevant state. For a ball in 3D space, position (x, y, z) and velocity components specify the state; for a pendulum, the angle and angular velocity suffice under simplifying assumptions. The choice of state variables is an abstraction decision: a neuron’s computational model focuses on electrical properties while ignoring other biochemical details, whereas a biochemist might do the reverse.
Differential equations then formalize how state variables change over time. Using a bacteria population in a Petri dish as an example, the growth rate is proportional to the current population: the rate of change dN/dt equals K·N, where K depends on conditions like temperature and food. The derivative is interpreted geometrically as the slope of the tangent to the population-vs-time curve. Real modeling, however, requires numerical methods rather than idealized infinitesimal changes: replace d with Δ and update the population in finite time steps. Measuring every five minutes instead of continuously assumes the rate stays roughly constant over that interval, enabling step-by-step predictions. This numerical approach matters because most differential equations—especially those relevant to neurons—lack closed-form solutions, so computation becomes the practical route.
Numerical solving also introduces tradeoffs and uncertainty. Smaller time steps improve accuracy but demand more computation. Parameter values like K are often unknown and must be inferred from data, meaning multiple simulations may be needed to fit observations, with finite precision inevitably leaving errors.
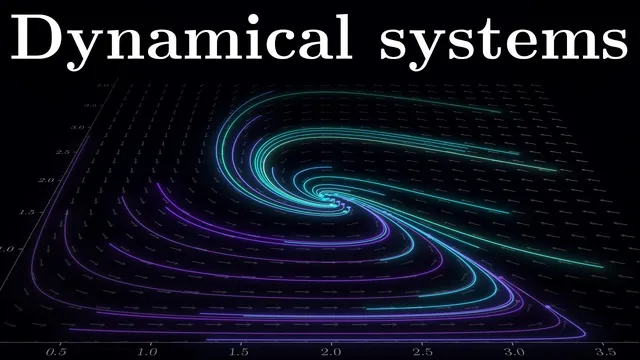
To build intuition for richer dynamics, the discussion shifts to coupled differential equations via a predator-prey model (rabbits and foxes). Rabbits grow proportionally to their population but decline when eaten by foxes, producing an interaction term −bxy. Foxes increase by converting consumed rabbits into reproduction (+cxy) but decline naturally without prey (−dy). Plotting time-series curves is useful, but the key geometric move is phase space: axes represent the two populations (x and y), while arrows at each point show the instantaneous direction of change. Equilibrium points occur where both derivatives vanish; one equilibrium is the trivial extinction state (0,0), while another represents a balance between predators and prey.
Around the extinction point, small perturbations drive the system away in one direction and toward extinction in another, signaling instability. Near the nonzero equilibrium, trajectories can form a closed loop—a limit cycle—meaning oscillations arise without explicitly inserting periodic functions. Changing assumptions can also transform behavior: limiting rabbit food can collapse the system from many possible cycles into a single stable equilibrium where trajectories converge.
These same geometric ideas—stability, equilibria, and limit cycles—are presented as the conceptual bridge to neuronal dynamics, where state variables may correspond to membrane potential and ion-channel states. The next step is promised as a dive into cellular biophysics to connect the math directly to how neurons generate spiking, bursting, and adaptation.
Cornell Notes
The core idea is that neurons can be treated as dynamical systems: their behavior at a moment depends on their state history, not just instantaneous input. Differential equations describe how state variables change over time, and numerical methods approximate derivatives using finite steps (Δ) because most realistic equations lack closed-form solutions. A bacteria-growth example turns the rule “growth rate is proportional to current population” into a differential equation and then into a step-by-step computation. To understand complex behavior without heavy calculation, the predator-prey model uses phase space, equilibrium points, and phase portraits; it can produce limit cycles—persistent oscillations emerging from interaction terms alone. These geometric concepts set up later applications to neuronal spiking and bursting.
What makes a dynamical system “fully described” at a given time, and how do state variables fit in?
How does the bacteria example turn “growth” into a differential equation, and what does the derivative mean geometrically?
Why switch from d to Δ in numerical methods, and what does that change in practice?
What is phase space, and how does it help reveal behavior without plotting time on the axes?
How do equilibrium points and limit cycles arise in the predator-prey model?
What role do parameter changes play in the qualitative dynamics of the predator-prey system?
Review Questions
- In the bacteria-growth model dN/dt = K·N, what does K represent, and how would changing the time step (e.g., from 5 minutes to 1 minute) affect numerical accuracy and computation cost?
- In phase space for the predator-prey model, what conditions define an equilibrium point, and how can stability be inferred from the direction of arrows near that point?
- Why can limit cycles appear without any explicit oscillatory functions in the equations, and what determines which limit cycle a system follows?
Key Points
- 1
Neurons are modeled as dynamical systems whose present behavior depends on their evolving state over time, not just instantaneous input.
- 2
State variables are the chosen real-number quantities that fully determine the system’s relevant state at a moment; what counts as “relevant” depends on the modeling abstraction.
- 3
Differential equations relate a quantity to its rate of change, with derivatives interpretable as tangent slopes on time-series graphs.
- 4
Numerical methods replace infinitesimal changes (d) with finite steps (Δ), enabling simulation when closed-form solutions are unavailable.
- 5
Smaller time steps improve accuracy but increase computational cost, and unknown parameters must often be fitted from data, introducing finite-precision uncertainty.
- 6
Phase portraits translate coupled differential equations into geometric flow in phase space, making stability and long-term behavior visible without exhaustive computation.
- 7
In the predator-prey model, equilibrium points and limit cycles emerge from interaction terms alone, and added constraints (like limited food) can switch the system from oscillations to convergence.